You Approved the Change.
But Do You Know What Else Changed?
In this QMS software demo, you’ll see how qmsWrapper connects CAPA, risk, and your Technical File — and what actually changes across your QMS before implementation.
Most QMS tools help you approve a change.
But they don’t show you what else it affects.
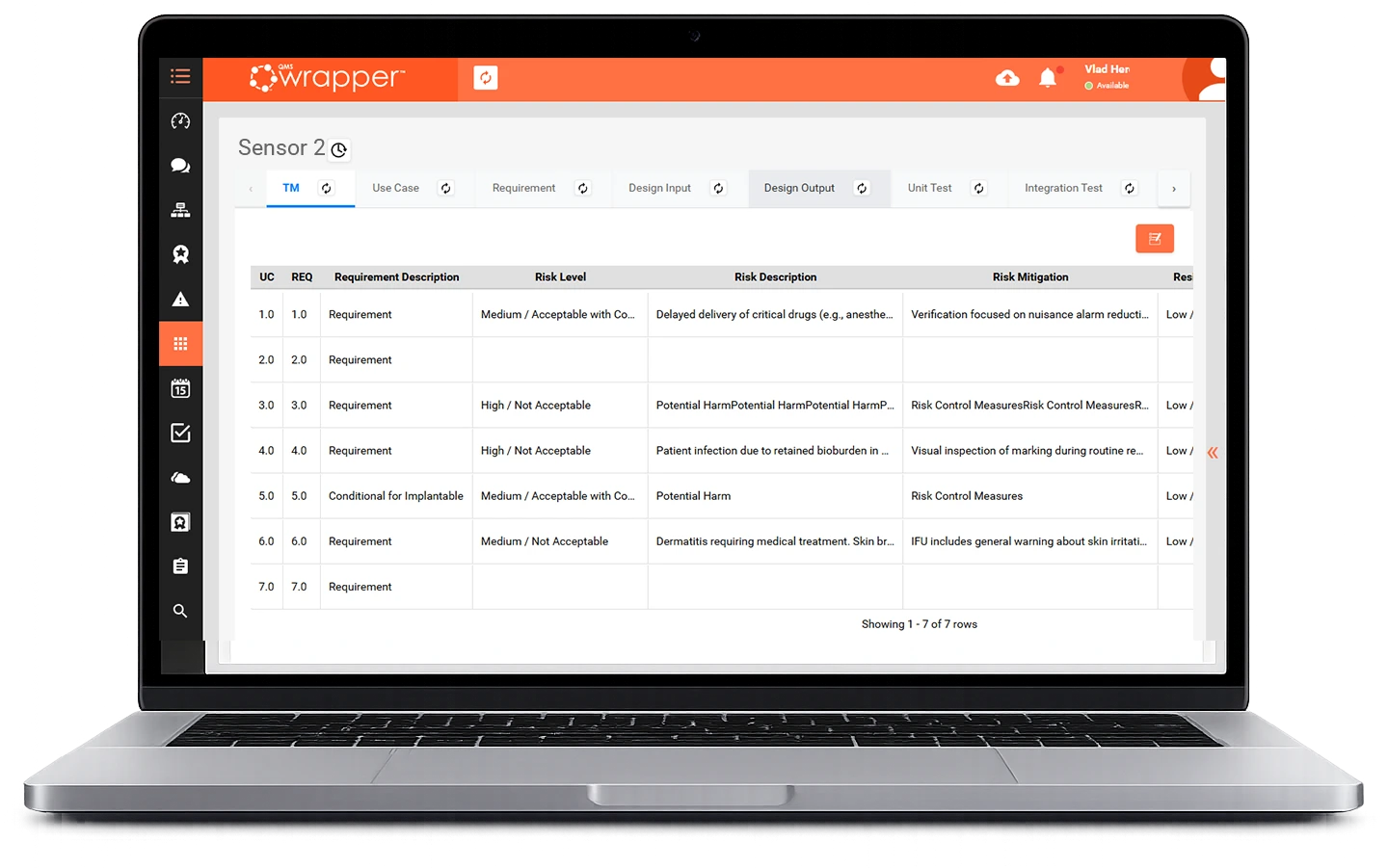
A requirement shifts.
A risk becomes outdated.
A test no longer applies.
That’s exactly what we show in this QMS software demo.
What You’ll See in the Demo
- How one event triggers impact across requirements, risk, and tests
- How Wrapper-Mapper identifies what must change
- How updates stay aligned across your Technical File
- How traceability is maintained automatically
- How AI-assisted workflows reduce manual work without losing control
Why MedTech Teams Choose qmsWrapper
- No more manual impact analysis across disconnected documents
- No more traceability gaps between CAPA, risk, and design
- No more audit surprises caused by missed updates
Everything is connected. Everything is traceable.
Built for MedTech SMEs — Not Enterprise Bureaucracy
Replace disconnected spreadsheets and rigid enterprise tools with one structured system for quality, risk, and design control.
AI-assisted workflows help teams prepare for audits faster — while keeping all decisions fully human and controlled.
- Validated for:
ISO 13485 • FDA 21 CFR Part 11 • ISO 14971 • EU MDR - Built for:
Medical Device & SaMD Teams - Includes:
eSignatures, Audit Trail, AI-Smart Forms, and full Traceability Matrix
See How Change Impact Works in Your Own QMS
Prefer to book directly? Schedule a time here.
Schedule Your Personalized qmsWrapper Demo
See how a Medical Device QMS works in real regulatory scenarios.
What Teams Want to Know Before Exploring a QMS Software Demo
What is a QMS software demo?
A QMS software demo is a walkthrough of how a quality management system works in real scenarios. In this QMS software demo, you can see how CAPA, risk, and Technical File data are connected and how changes impact the system before implementation.
What should I look for in a QMS software demo?
In a QMS software demo, you should look for how traceability is maintained, how changes affect related records, and whether CAPA, risk, and design data stay connected. It should reflect real workflows, not just static features.
Who is this QMS software demo for?
This QMS software demo is designed for medical device companies and Software as a Medical Device (SaMD) teams that need structured, audit-ready quality processes. It is especially relevant for startups and small to medium-sized companies that want to implement a compliant QMS without enterprise complexity.
How much does qmsWrapper cost?
qmsWrapper starts at $6,000 per year, with all modules included — including CAPA, risk management, design control, document control, and AI-assisted workflows. There are no add-ons or hidden modules.
Is everything included in the QMS software demo and pricing?
Yes. This QMS software demo reflects the full system. All modules are included in the pricing, including AI-assisted features, traceability, CAPA, risk management, and Technical File control.
How long does implementation take after the demo?
Most MedTech teams start using qmsWrapper within weeks. The system is pre-configured for regulated workflows, so there is no need for long implementation or customization projects.
Is this QMS software demo relevant for FDA and ISO 13485 compliance?
Yes. This QMS software demo shows workflows aligned with FDA and ISO 13485 requirements, including traceability, CAPA, risk management, and audit-ready records.